1. 引言
次氯酸(ClO−)和过氧化亚硝酸盐(ONOO−)作为生命体中的重要活性氧 [1] [2] [3] [4] [5],具有较强氧化性和亲核性 [6] [7],在维持体内氧化还原平衡中发挥了重要作用,但过量活性氧也会引起疾病 [8] [9],因此开发准确识别活性氧的有效方法具有重要的意义。与其他检测方法相比,荧光检测法具有灵敏度高、选择性好、实时检测等优点 [10] [11] [12] [13]。人们已经开发出形式多样的识别活性氧/氮的荧光探针。到目前为止,基于酰胺/酯的氧化裂解 [14] [15] [16],双键的氧化裂解(C=C、C=N、C=S和N=N) [17] [18] [19],硫醚化合物/硒化合物的氧化 [20] 等作用原理开发设计了许多ClO−荧光探针;同时基于硼酸盐/硼酸氧化裂解 [21] [22] [23] 成苯酚,与反应酮生成二环氧乙烷 [24] [25],有机硒/有机碲化合物的氧化 [26] [27] [28],芳香族化合物的硝化 [29],苯酚的氧化 [30],亚硝胺 [31] 的生成及其水解的作用机制设计合成了ONOO−的荧光探针,上述探针均对ClO−和ONOO−的识别表现出较高的灵敏度和选择性,但在探针合成上也存在识别基构建困难,不易合成等缺点,而酰胺键易于合成,往往通过羧酸衍生物氨解既可以快速合成,因此利用酰胺键水解前后的荧光信号改变设计次氯酸或过氧化亚硝酸盐荧光探针具备可行性和必要性。
罗丹明荧光母体螺内酰胺闭环时溶液为无色、荧光强度较弱,而开环结构具有光稳定性好、刚性共轭结构大、发射波长长、水溶性好的优势,往往用于可视化荧光探针的设计与合成。而四苯基乙烯荧光母体作为聚集诱导发光(AIE)物质的典型代表,具有结构易修饰等优势,鉴于上述荧光母体优势,本文利用2,6-吡啶二甲酰氯通过酰胺键连接四苯基乙烯和罗丹明结构,合成得到探针TPE-RhB,探针充分发挥罗丹明的可视化优势和四苯基乙烯的聚集诱导发光特性,利用ClO−和ONOO−氧化和亲核能力的差异,通过对探针TPE-RhB中酰胺键识别机制的不同,同时实现了对ClO−和ONOO−的可视化选择性识别。这项研究可以扩大检测活性氧ClO−和ONOO−荧光探针的设计思路,为众多的研究者设计合成荧光探针提供依据。
2. 实验部分
2.1. 仪器与试剂
通过美国Varian公司Varian 400-MR测定核磁共振数据;由日立U-3310紫外可见分光光度计测定紫外吸收光谱数据;利用Bruker TENSOR27红外光谱仪测定红外光谱数据;借助Varian Cary Eclipse荧光分光光度计测定荧光光谱数据;由TRACE MS质谱仪测定质谱数据。
无水乙醇,四氢呋喃,2,6-吡啶二甲酰氯等均为市售分析纯或化学纯试剂。
2.2. 化合物TPE-RhB的合成
参照图式1合成探针TPE-RhB:称量0.306 g (1.5 mmol) 2,6-吡啶二甲酰氯置于100 mL干燥圆底烧瓶中,并用15.0 mL二氯甲烷溶解,启动搅拌,将体系冰浴降温到0℃左右后,缓慢滴入0.375 g (1 mmol) 1,1-二(4-甲基苯基)-2-(4-氨基苯基)-2-苯基乙烯(CH3-TPE-NH2)的二氯甲烷溶液。待CH3-TPE-NH2基本消失后滴入0.91 g (2 mmol)罗丹明酰肼(RhB-NH2)的二氯甲烷溶液,全部滴加完毕后,加入0.5 mL三乙胺,缓慢回至室温,30 min停止反应,加入20 mL蒸馏水猝灭反应,二氯甲烷萃取(3 × 30 mL)有机相,并用无水硫酸镁干燥2 h,过滤、洗涤、旋蒸溶剂得到紫红色粗品,粗品用石油醚/乙酸乙酯(v/v = 3:1)柱层析分离得白色粉末固体TPE-RhB 0.5 g,产率:58%,m.p. 249℃~251℃;表征数据如图S4~S6,1H NMR (CDCl3,400 MHz) δ:1.05 (t, J = 7.2 Hz, J = 7 Hz, 12H),2.24 (s, 6H),3.11~3.24 (m, 8H),6.22 (t, J = 8.4 Hz, 2H),6.23 (s, 2H),6.61 (d, J = 8.8 Hz, 2H),6.85~7.14 (m, 16H),7.33~7.5 (m, 5H),7.66 (s, 2H),7.78 (d, J = 6.8 Hz,1H),8.23 (d, J = 7.2 Hz, 1H),9.68 (s, 1H);13C NMR (CDCl3, 100 MHz) δ:12.5,21.1,021.2,44.2,97.8,107.9,119.61,123.8,124.0,126.0,127.5,128.2,128.4,129.1,131.1,131.2,131.4,131.6,133.0,135.8,136.0,138.3,139.4,139.8,140.6,140.8,141.0,144.46,147.1,148.8,149.0,151.6,153.8,160.6,160.8,166.2. IR (v, KBr):2970,1633,1518,1402,1221,1119,699 cm−1;ESI-MS m/z (%):964.9 (M + H 100)。
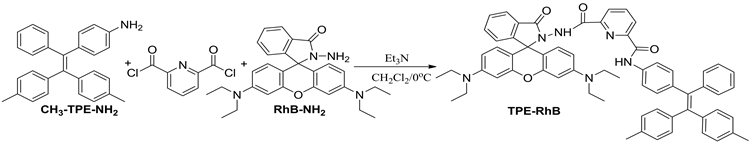
Scheme 1. Synthetic routes of probe TPE-RhB
图式1. 探针TPE-RhB的合成路线
2.3. 溶液配制方法
称量48.15 mg探针TPE-RhB置于干燥的小烧杯中,用无水乙醇溶解,并转移至100毫升容量瓶中,定容得浓度为5.0 × 10−4 mol/L储备液。识别ClO−和ONOO−的光谱研究均在EtOH/H2O (50 μmol/L, v/v, 1/1,PBS, pH 7.4)溶液中进行,激发波长为λEx = 364 nm,激发和发射狭缝宽度均为5 nm。
活性氧/氮的配制参照文献方法 [32] [33]:NaClO,H2O2,叔丁基过氧化氢(TBHP),NO均用超纯水进行稀释得到;·OH通过Fe2+和H2O2发生芬顿反应得到;TBO·通过Fe2+和TBHP发生芬顿反应得到;1O2通过钼酸钠和H2O2发生反应得到;ONOO−通过以下方法获得:冰浴条件下,HCl水溶液(0.6 M, 5 mL)和H2O2溶液(0.7 M, 5 mL)混合,搅拌20 min后,加入NaOH溶液(1.5 M, 5 mL)和NaNO2溶液(0.6 M, 5 mL)的混合溶液继续反应1 min,得到微黄溶液,即为ONOO−储备液,储备液在−20℃下保存,其浓度通过紫外–可见吸收光谱由302 nm处的吸光度计算,ε = 1670 M−1 cm−l。
3. 结果与讨论
3.1. 聚集诱导荧光性质研究
众所周知,四苯基乙烯衍生物是典型的AIE物质,当连接上罗丹明荧光母体后是否保留该特性,对能否利用聚集诱导发光原理设计探针具有重要意义。
结果如图1所示,化合物TPE-RhB的荧光强度随含水量的增加呈现上升趋势,当水体积分数为80%时,荧光强度达到最大为757,比纯乙醇溶液时增强了37倍,结果表明当四苯乙烯与罗丹明连接后探针TPE-RhB仍具有典型的AIE特征。
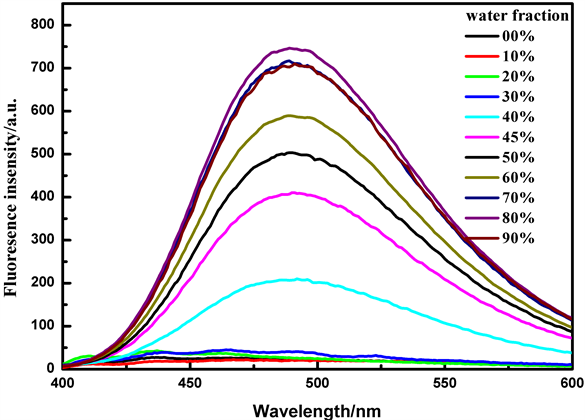
Figure 1. Emission spectra of probe TPE-RhB (50 μmol/L) in EtOH-H2O mixtures with different water fraction (λEm = 490 nm)
图1. 探针TPE-RhB (50 μmol/L)在不同水体积分数的乙醇/水溶液中的聚集诱导荧光图(λEm = 490 nm)
3.2. 探针TPE-RhB的选择性识别研究
为考察TPE-RhB对活性氧的响应情况,测定了探针TPE-RhB与活性氧反应前后的紫外光谱和荧光光谱,结果如图2和图S1~S3所示。
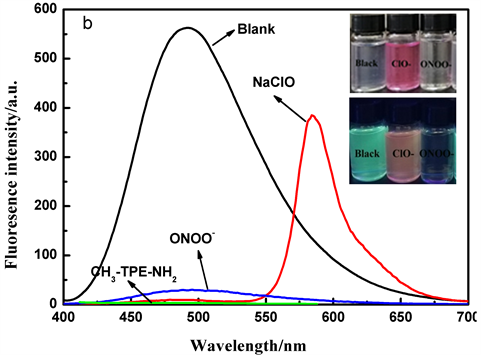
Figure 2. Uv-visible absorption spectrum (a) and fluorescence spectra (b) of probe TPE-RhB solution after adding 8 equiv. ClO− and ONOO− (the inner pictures were taken under visible light and 365 nm uv lamps before and after adding ClO− and ONOO− into the probe solution)
图2. 探针TPE-RhB的乙醇/水溶液中加入8 equiv. ClO−和ONOO−后的紫外–可见吸收光谱图(a)和荧光光谱图(b) (内图为探针溶液中加入ClO−、ONOO−前后在可见光和365 nm紫外灯下照片)
从上述结果中可知,探针TPE-RhB的乙醇/水溶液在325 nm和360 nm处有明显的紫外吸收峰,在加入ClO−后,该位置的紫外吸收略微减弱,同时582 nm处产生新的吸收峰。但与ONOO−反应后,探针在325 nm和360 nm的紫外吸收峰明显增强,说明探针与ONOO−、ClO−发生了化学反应(图2(a))。探针在加入ClO−后,在5秒内引起探针溶液颜色由无色变为粉红色,同时490 nm处发生猝灭比为98.6%的荧光猝灭效应,而582 nm处实现了荧光增强,I582/I490增大了218倍,实现对ClO−的可视化荧光识别。当加入ONOO−后,30秒内仅观察到490 nm处荧光强度急剧下降,猝灭比为97.5% (图2(b))。而在其他活性氧或金属离子存在下则无上述现象,表明探针对ONOO−、ClO−表现出可视化识别能力。
3.3. 探针TPE-RhB识别ClO−的光谱性质研究
3.3.1. 探针TPE-RhB识别ClO−前后在不同pH条件下荧光性质研究
研究了pH值对该探针的荧光响应影响,结果如图3所示。
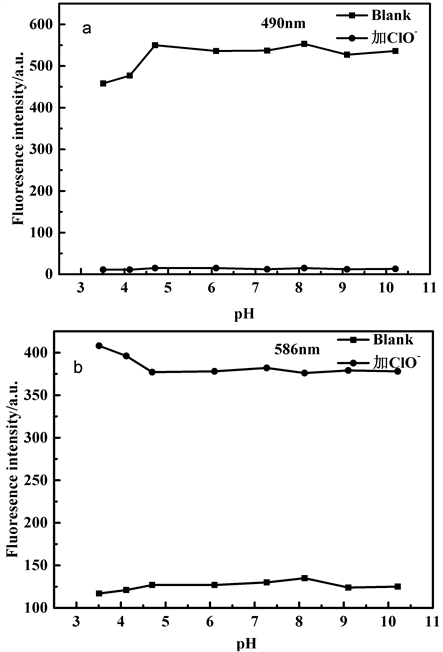
Figure 3. The pH value influence on the fluorescence intensity of probe TPE- RhB in the absence and presence of ClO− (a: λEm = 490 nm, b: λEm = 586 nm)
图3. 在不同pH值条件下,探针TPE-RhB与ClO−作用前后的荧光强度变化图(a: λEm = 490 nm, b: λEm = 586 nm)
在无HClO/ClO−存在下,探针溶液在pH 4.7~10.2范围内表现出较为稳定的荧光响应,且始终处于无色状态。在HClO/ClO−存在下,探针溶液在pH 3.5~10.2范围内,490 nm荧光强度表现出猝灭,586 nm出现新的发射峰且溶液变为粉红色,由于生命体系大部分为中性,在后续光谱性质测试中以PBS溶液为缓冲溶液,pH值选为7.4。
3.3.2. 探针TPE-RhB与ClO−反应动力学实验
在探针TPE-RhB的乙醇/水加入8 equiv. ClO−,观察并测定探针溶液颜色以及荧光强度变化,结果如图4所示。
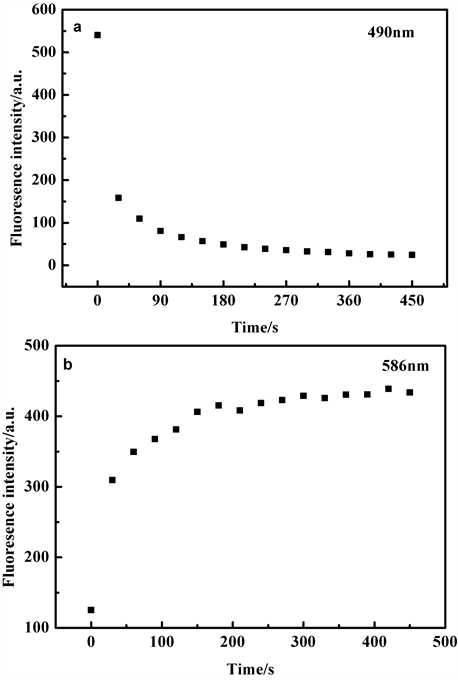
Figure 4. Time-dependent fluorescence changes of probe TPE-RhB in the absence and presence of ClO− (a: λEm = 490 nm, b: λEm = 586 nm)
图4. 在不同反应时间条件下,探针TPE-RhB与ClO−作用前后的荧光强度变化图(a: λEm = 490 nm, b: λEm = 586 nm)
当探针TPE-RhB的乙醇/水溶液加入8 equiv. ClO−后,探针溶液5秒内迅速变红,同时490 nm处荧光强度迅速降低,而586 nm处的荧光强度迅速增加,在180 s左右,F586荧光强度保持稳定,结果表明探针与ClO−反应非常迅速,后续荧光测试选择180秒后测定。
3.3.3. 探针TPE-RhB在不同浓度ClO−存在下的荧光滴定实验
利用荧光光谱法测定了在不同浓度ClO−存在下的荧光发射强度,结果如图5所示。
在探针TPE-RhB的乙醇/水溶液中,490 nm处荧光强度逐渐减弱,其荧光强度与ClO−浓度在30~250 μM范围符合线性方程y = −1.91 × 106x + 566.90,线性系数为0.9919 (图5(b)),通过公式D = 3Sd/ρ (其中ρ是荧光强度与ClO−的斜率,Sd是空白标准偏差),得出检测限为8.2 × 10−6 M,586 nm发射波长下荧光强度与ClO−浓度在300~500 μM范围内符合线性方程y = 1.50 × 106x − 375.14,线性系数为0.9812 (图5(c)),并通过公式D = 3Sd/ρ (其中ρ是荧光强度与ClO−的斜率,Sd是空白标准偏差),得出检出限为6.1 × 10−6 M。该法已应用于自来水中ClO−的加标回收定量检测,回收率均在97%~102% (表S1和表S2,研究结果表明了该法的有效性)。
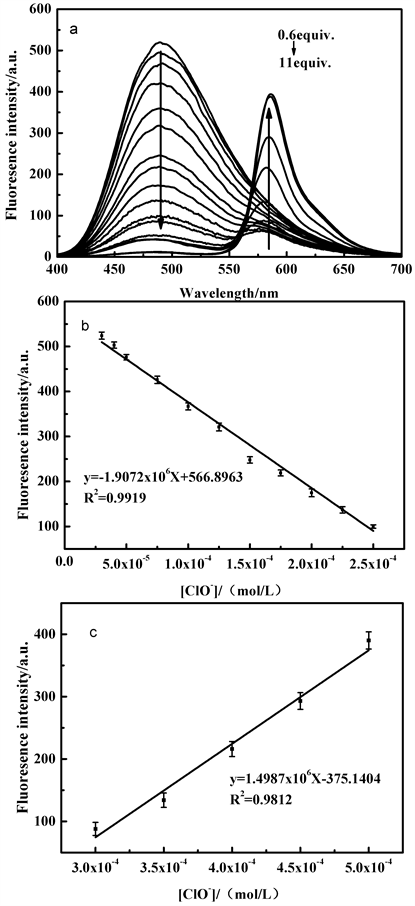
Figure 5. Fluorescence emission spectra (a) and linear relationship of probe TPE-RhB in the presence of different ClO− concentrations (b: λEm = 490 nm, c: λEm = 586 nm)
图5. 在不同浓度ClO−存在下探针TPE-RhB溶液的荧光发射光谱图(a)及线性关系图(b: λEm = 490 nm, c: λEm = 586 nm)
3.3.4. 探针TPE-RhB与ClO−识别机理初探
为了解TPE-RhB与ClO−的作用机制,对TPE-RhB-ClO−混合物进行了ESI-MS分析,结果如图6所示,初步将m/z = 443.7和m/z = 527.76处的峰分别归因于3和6,因此提出了图7中描述的识别机制。即探针TPE-RhB经NaClO氧化诱导罗丹明结构开环得到罗丹明N-Cl化合物1,并继续脱氯化氢获得偶氮化合物2,2经水解获得罗丹明B 3和中间体4,4进一步水解脱氮得化合物6。在这识别过程中,化合物3的产生将会导致586 nm处的荧光强度迅速增加,并伴随探针溶液颜色变红,而化合物6在溶液中溶解性更好,则会引起490 nm荧光猝灭。
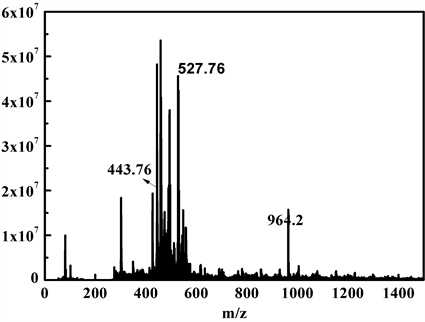
Figure 6. ESI-MS diagram of TPE-RhB-ClO− mixture
图6. TPE-RhB-ClO−混合物的ESI-MS图
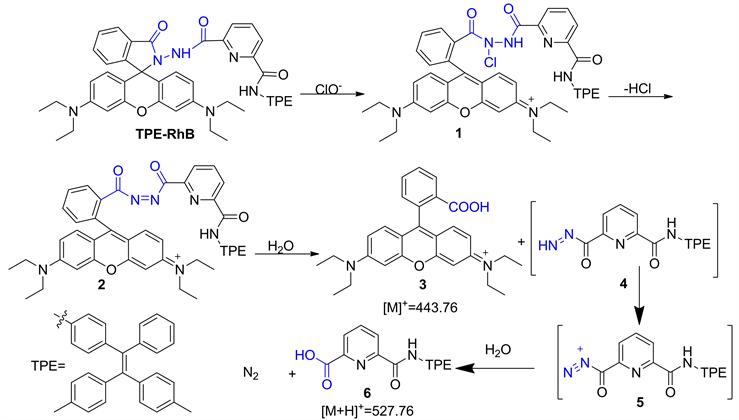
Figure 7. The possible mechanism of probe TPE-RhB identifies ClO−
图7. 探针TPE-RhB识别ClO−的可能机理
3.4. 探针TPE-RhB识别ONOO−的光谱性质研究
3.4.1. 探针TPE-RhB识别ONOO−前后在不同pH条件下荧光性质研究
研究了不同pH值条件下该探针对ONOO−的荧光响应,结果如图8所示。显然,在无ONOO−存在下,探针溶液在pH 3.5~10.2范围内荧光强度较为稳定;在ONOO−存在下,pH 3.5~10.2范围内的,490 nm荧光强度表现出猝灭,探针溶液由无色聚集状态变为无色透明状态。由于生命体系大部分为中性,在后续光谱性质测试中以PBS溶液为缓冲溶液,pH值选为7.4。
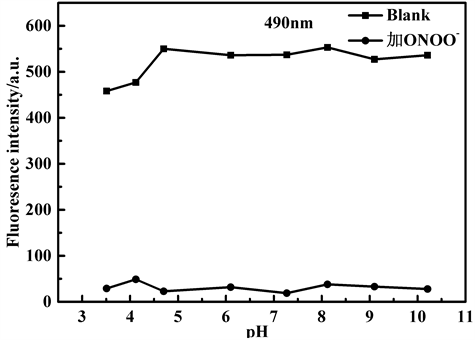
Figure 8. The pH value influence on the fluorescence intensity of probe TPE-RhB in the absence and presence of ONOO− (λEm = 586 nm)
图8. 在不同pH值条件下,探针TPE-RhB与ONOO−作用前后的荧光强度变化图(λEm = 586 nm)
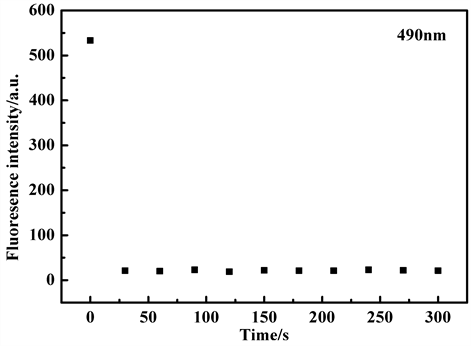
Figure 9. Time-dependent fluorescence changes of probe TPE-RhB in the absence and presence of ONOO− (λEm = 490 nm)
图9. 在不同反应时间条件下,探针TPE-RhB与ONOO−作用前后的荧光强度变化图(λEm = 490 nm)
3.4.2. 探针TPE-RhB与ONOO−反应动力学测试
为了获得最佳的灵敏度,对响应时间也进行了优化,在加入ONOO−后,结果如图9表明,在30秒内观察到荧光强度急剧下降,这表明探针与ONOO−反应非常迅速。这种快速反应的探针分子可作为实时检测ONOO−的有效探针,并优于许多其他探针。
3.4.3. 探针TPE-RhB在不同浓度ONOO−存在下的荧光滴定实验
在不用浓度ONOO−存在下,探针TPE-RhB溶液仅在490 nm处荧光强度逐渐减弱,结果如图10所示。
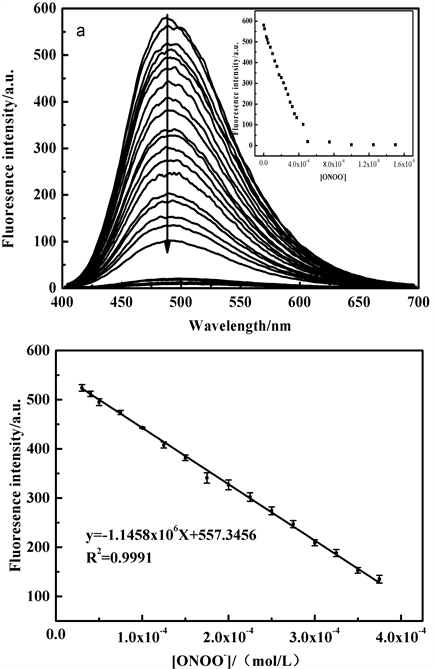
Figure 10. Fluorescence emission spectra (a) and linear relationship (b: λEm = 490 nm) of probe TPE-RhB in the presence of different ONOO− concentrations
图10. 探针TPE-RhB的乙醇/水溶液中逐渐加入不同浓度的ONOO−后的荧光光谱图(a)及线性关系曲线(b: λEm = 490 nm)
从上图可知,荧光强度与ONOO−浓度在30~375 μM范围内符合线性方程y = −1.15 × 106x + 557.35,线性系数为0.9991,并通过公式D = 3Sd/ρ得出检出限为1.43 × 10−5 M,说明该探针可应用于ONOO−的定量检测。
3.4.4. 探针TPE-RhB与ONOO−反应识别机理初探
为了解TPE-RhB与ONOO−之间的作用机制,对TPE-RhB-ONOO−混合物进行了ESI-MS分析,结果如图11所示,初步将m/z = 363.9和m/z = 302.1处的峰归因于探针水解产物CH3-TPE-NH2脱甲基和脱4-甲苯基后产生的碎片离子峰。因此提出了图12中描述的信号机制,即在ONOO−作用下,增强水分子的亲核性,引起TPE-RhB结构中四苯基乙烯侧的酰胺键优先水解,获得水解产物TPE-NH2,诱导荧光猝灭,但罗丹明荧光母体在此过程中未产生诱导开环,因此探针溶液颜色和586 nm处荧光强度均未发生变化。
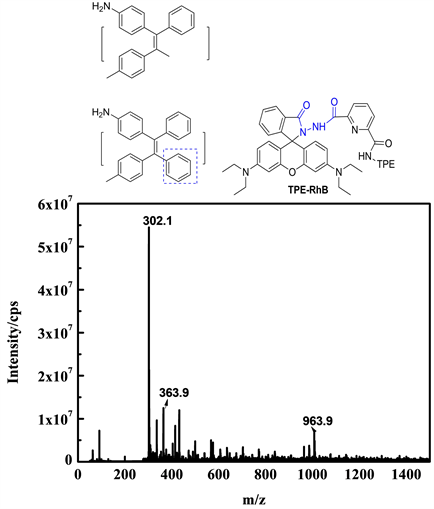
Figure 11. ESI-MS diagram of TPE-RhB-ONOO− mixture
图11. TPE-RhB-ONOO−混合物的ESI-MS图
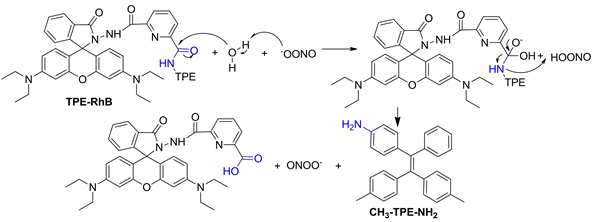
Figure 12. The possible mechanism of probe TPE-RhB identifies ONOO−
图12. 探针TPE-RhB识别ONOO−的可能机理
4. 结论
本文利用2,6-吡啶二甲酰氯合成得到基于四苯乙烯–罗丹明的双酰胺化合物TPE-RhB,通过光谱性能研究表明,探针TPE-RhB在5秒内实现可视化比率荧光识别ClO−,探针溶液由无色变为粉色,490 nm处荧光产生98.6%的猝灭,在582 nm处实现了荧光增强。而在ONOO−存在时,30秒内仅观察到490 nm处荧光强度下降,猝灭比为97.5%,对其它活性氧/氮及金属离子则无明显变化。通过TPE-RhB-ClO−和TPE-RhB-ONOO−混合物的ESI-MS分析,初步证明在ClO−作用下,TPE-RhB结构中罗丹明侧的酰胺键优先氧化水解,引起探针溶液荧光和颜色多重信号变化,但ONOO−存在时,则优先引起四苯乙烯侧的酰胺键水解,仅实现490 nm处的荧光变化。
基金项目
新疆维吾尔自治区高校科研计划自然科学基金项目(XJEDU2019Y028)和新疆师范大学优秀青年教师科研启动基金项目(XJNU202013)资助。
附件支持信息

Figure S1. Photos of the probe TPE-RhB in ethanol/water (50 μmol/L, v/v, 1/1, PBS, pH 7.4) after adding different ROS/RON and metal ions (8 equiv.) under natural light
图S1. 探针TPE-RhB的乙醇/水(50 μmol/L, v/v, 1/1, PBS, pH 7.4)中加入不同ROS/RON及金属离子(8 equiv.)后的在日光灯下照片

Figure S2. Photos of probe TPE-RhB under 365 nm UV lamp after adding ROS/RON and metal ions (8 equiv.) to ethanol/water (50 μmol/L, v/v, 1/1, PBS, pH 7.4)
图S2. 探针TPE-RhB的乙醇/水(50 μmol/L, v/v, 1/1, PBS, pH 7.4)中加入不同ROS/RON及金属离子(8 equiv.)后的在365 nm紫外灯下照片
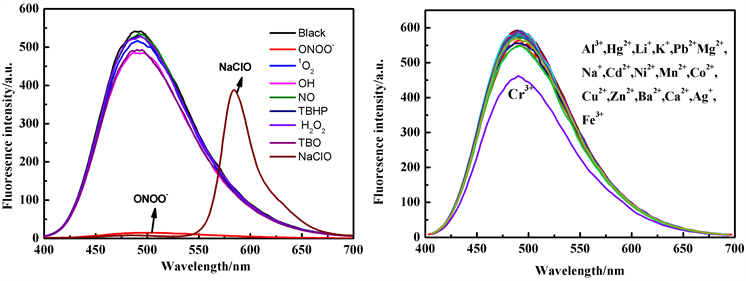
Figure S3. Fluorescence spectra (Ex = 364 nm) of probe TPE-RhB after adding different ROS/RON and metal ions (8 equiv.) to ethanol/water (50 μmol/L, v/v, 1/1, PBS, pH 7.4)
图S3. 探针TPE-RhB的乙醇/水(50 μmol/L, v/v, 1/1, PBS, pH 7.4)中加入不同ROS/RON及金属离子(8 equiv.)后的荧光光谱图(Ex = 364 nm)
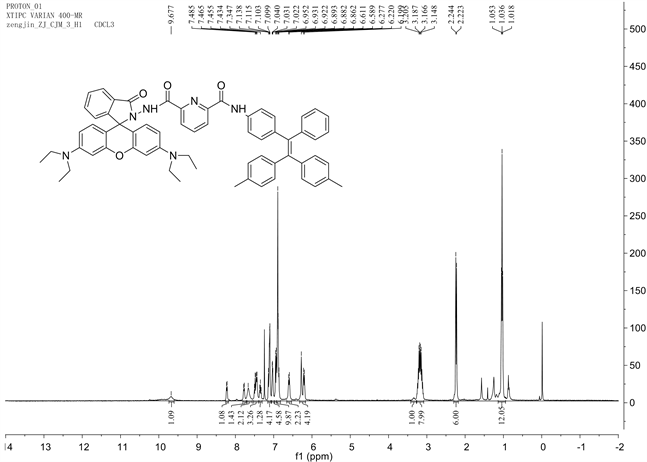
Figure S4. 1HNMR diagram of compound TPE-RhB
图S4. 化合物TPE-RhB的1HNMR图
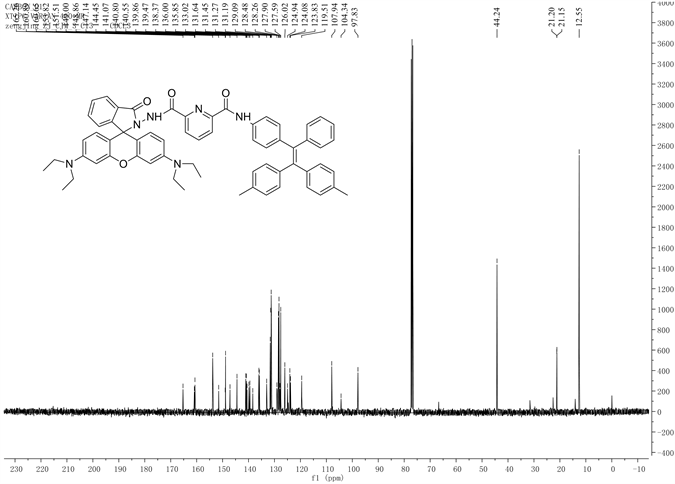
Figure S5. 13CNMR diagram of compound TPE-RhB
图S5. 化合物TPE-RhB的13CNMR图
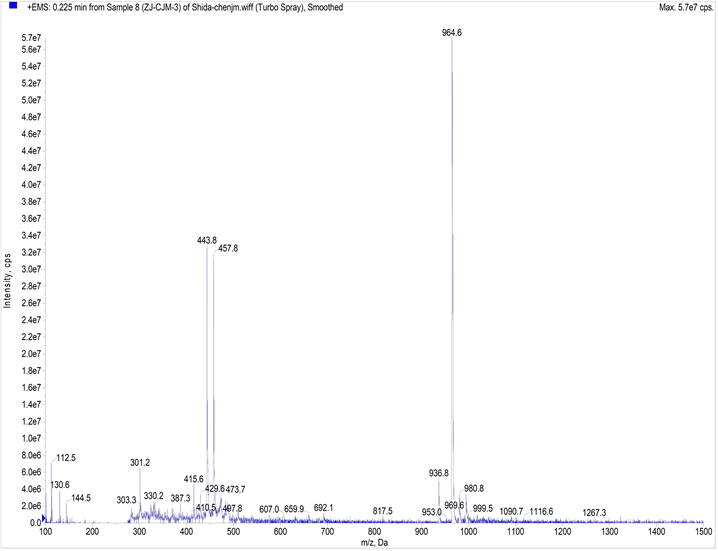
Figure S6. ESI-MS diagram of compound TPE-RhB
图S6. 化合物TPE-RhB的ESI-MS图

Table S1. Recovery of ClO− in water samples measured by probe at 490 nm
表S1. 490 nm处探针测定水样中ClO−的回收率研究

Table S2. Recovery of ClO− in water samples measured by probe at 586 nm
表S2. 586 nm处探针测定水样中ClO−的回收率研究
NOTES
*通讯作者。