1. 引言
黄铁矿在地下沉积物中含量丰富,广泛分布于江河海口和地下岩层中。尽管黄铁矿在缺氧环境中形成,但在有氧环境中仍然稳定 [1] 。黄铁矿对锑(III)的氧化 [2] ,铬(VI)的还原 [3] ,三氯乙烯(TCE) [4] 、双氯芬酸 [5] 、硝基苯 [6] 、氯苯胺 [7] 和有机染料 [8] 等有毒有害物质的迁移、转化和降解有着重要的影响。黄铁矿因其表面活性、晶体结构等特性,具有较好的吸附作用,能通过吸附作用去除水中不同浓度磷 [9] 、硒酸根 [10] 、六价铬 [11] 以及难降解染料 [12] 。
本研究以黄铁矿为吸附剂,探究了吸附时间、pH和温度对黄铁矿吸附RhB与SRB过程的影响。利用Langmuir和Freundlich吸附等温线模型、准一级和准二级动力学模型分别对吸附过程进行拟合,并结合热力学计算对黄铁矿吸附RhB和SRB的机理进行了初步比较探讨。最后对吸附后的黄铁矿进行煅烧处理后再次进行吸附实验,结果表明黄铁矿不具备多次重复利用的价值,可以直接进行回收填埋或用作工业建筑材料。本研究为利用黄铁矿处理印染废水、改善水环境提供依据。
2. 实验部分
2.1. 主要仪器及试剂
主要仪器:紫外可见分光光度计(V-1600BPC,上海美谱达仪器有限公司),落地冷冻摇床(HZ-9310KA,常州恒隆仪器有限公司)。
主要试剂:实验用黄铁矿购自铜陵市达业进出口有限公司,用铁锤将块状黄铁矿捶碎,随后进一步粉碎,收集粉末过100目筛,将黄铁矿粉末储存于密封收集袋中,做好标记以供实验使用。罗丹明B (RhB)、酸性桃红(SRB),结构见图1,5.0 × 10−4 mol/L水溶液作为标准溶液,稀释成不同浓度的水溶液进行实验;实验所用其他试剂均为分析纯,水为二次蒸馏水。
Figure 1. Structure of RhB (a) and SRB (b)
图1. RhB (a)和SRB (b)的结构
2.2. 实验方法
2.2.1. 标准曲线的绘制
吸取3 mL RhB标准贮备溶液于100 mL容量瓶中,用蒸馏水稀释至标线,得到1.5× 10−5 mol/L的RhB标准溶液,摇匀备用。
分别取0、1.5、3.0、4.5、6.0、7.5 mL RhB标准溶液于6支50 mL具塞比色管中,用蒸馏水定容,摇匀。此标准系列RhB的溶度分别为0、0.45 × 10−5、0.9 × 10−5、1.35 × 10−5、1.8 × 10−5、2.25 × 10−5 mol/L。用紫外可见分光光度计在波长554 nm下测定吸光度A,通过得到的数据绘制标准曲线。
SRB标准曲线的绘制方法同RhB,检测波长为565 nm。
2.2.2. 吸附实验
吸取将一定量的黄铁矿粉末和RhB溶液倒入500 mL锥形瓶中,在黑暗条件下振荡吸附,转速为180 r/min,每隔相同的时间取样3 mL于5 mL离心管中,3600 r/min高速离心5 min,取上清液于554 nm处测定吸光度值A,并记录数据。SRB的吸附实验同上。
根据公式(1)计算去除率(%),其中c0为初始浓度(mol/L),ce为吸附达到平衡时的浓度(mol/L)。
(1)
利用公式(2)计算t时间的吸附量qt (mg/g),其中qt为t时间的吸附量qt (mg/g);c0为初始浓度(mol/L);ct为t时间的浓度(mol/L);v为溶液的体积(mL);m为黄铁矿粉末的用量(g),并作t~qt图。对吸附进行准一级和准二级动力学模型模拟,并作
和t~t/qt图,qe和qt (mg/g)分别为吸附达到平衡和时间t时,吸附剂对底物的吸附量;k1是准一级动力学吸附速率常数(/h);k2是准一级动力学吸附速率常数(g/mmol/h),根据公式(3)和(4)计算动力学模型各参数。
(2)
(3)
(4)
利用公式(5)和(6)对吸附等温线实验数据分别进行Langumir和Freundlich吸附等温线模型拟合。
(5)
(6)
ce (mg/L)为吸附达到平衡时溶液中剩余的底物的浓度;qe (mg/g)是吸附达到平衡时底物被吸附的浓度;kL (L/g)和aL (L/mg)为Langmuir吸附等温线常数;kF为Freundlich吸附等温线常数(L/mg);1/nF是非均质性参数。
通过公式(7)可以计算出表观热力学函数
和
,其中Kd (L/mg)表示不同温度下的Langmuir吸附常数;R为理想气体常数8.314 J/(mol K);T为开尔文温度。利用公式(8)计算吉布斯自由能
。
(7)
(8)
3. 结果与讨论
3.1. 标准曲线的绘制
图2为RhB和SRB的浓度对应吸光度的标准曲线,RhB标准曲线的线性方程为
,R2 = 0.9997;SRB标准曲线的线性方程为
,R2 = 0.9995。
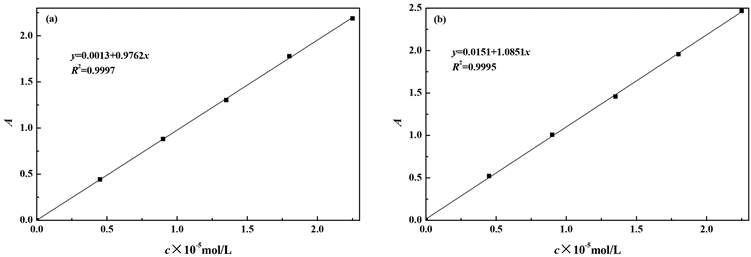
Figure 2. Standard curves of RhB (a) and SRB (b)
图2. RhB (a)和SRB (b)的标准曲线
3.2. 吸附动力学
黄铁矿粉末对RhB和SRB的吸附见图3。可以看出,前2 h黄铁矿粉末对RhB和SRB吸附速度很快,后面逐渐变慢,5 h后吸附达到平衡,此时黄铁矿粉末对RhB和SRB的最大吸附量分别为2.89和1.70 mg/g。后面实验中,都使黄铁矿粉末与RhB和SRB的接触时间达到5 h以使吸附达到平衡。
对实验得到的动力学数据进行准一级和二级动力学模型模拟,结果分别见图4(a)和图4(b),其动力学模型参数见表1。从结果中可以看出,黄铁矿粉末吸附RhB和SRB都符合准二级动力学模型,线性相关系数分别为0.9895和0.9876。根据线性图计算得到的最大饱和吸附量分别为3.04和1.78 mg/g,接近实验值。
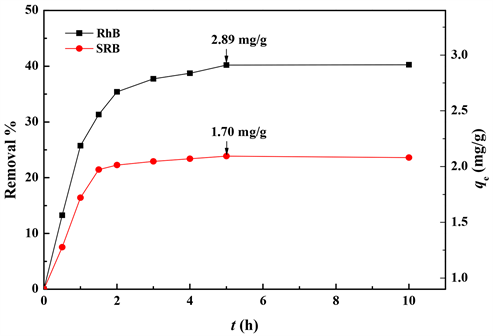 T = 20℃ ± 1℃;v = 50 mL;m = 25 mg;c0 = 1.5 × 10−5 mol/L;pH = 7 ± 0.1
T = 20℃ ± 1℃;v = 50 mL;m = 25 mg;c0 = 1.5 × 10−5 mol/L;pH = 7 ± 0.1
Figure 3. Adsorption kinetics
图3. 吸附动力学
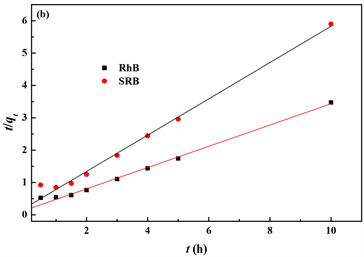
Figure 4. Plot of pseudo-first-(a) and pseudo-second-order rate (b) for the adsorption of RhB and SRB on pyrite powder
图4. 黄铁矿粉末吸附RhB和SRB的准一级(a)和准二级(b)动力学曲线

Table 1. Comparison of pseudo-first- and pseudo-second-order model parameters
表1. 黄铁矿粉末吸附RhB和SRB的准一级和准二级动力学模型参数的比较
3.3. 吸附等温线
对黄铁矿粉末吸附RhB和SRB的实验数据分别进行Langumir和Freundlich吸附等温线模型拟合,线性图见图5,吸附等温线模型拟合参数见表2。结果发现,黄铁矿粉末对RhB和SRB的吸附都与Langumir模型拟合程度更高,线性相关系数分别为0.9987和0.9995。Langmuir吸附模型相关系数R2大于Freundlich吸附模型的相关系数R2,表明黄铁矿吸附RhB和SRB更符合Langmuir吸附等温线模型,属于单分子层吸附。

Figure 5. Adsorption isotherm of RhB and SRB on pyrite powder
图5. 黄铁矿粉末吸附RhB和SRB的Langmuir (a)和Freundlich (b)吸附等温线

Table 2. Parameters for Langmuir and Freundlich sorption isotherms
表2. Langmuir和Freundlich吸附等温线参数
3.4. 热力学研究
热力学研究可以更全面的评价黄铁矿粉末对RhB和SRB的吸附过程,温度对黄铁矿粉末吸附RhB和SRB的影响见图6。
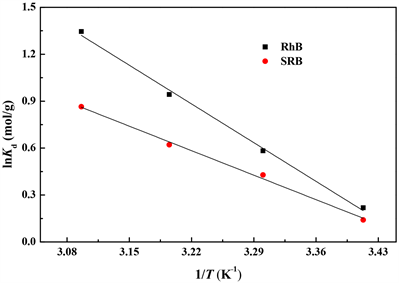
Figure 6. Effect of temperature on adsorption of RhB and SRB by pyrite powder
图6. 温度对黄铁矿粉末吸附RhB和SRB的影响
不同温度下的表观热力学常数见表3。黄铁矿粉末吸附RhB和SRB的
都为正值,表明黄铁矿粉末对RhB和SRB的吸附都是吸热过程。同时,温度越高,
越大,表明温度越高黄铁矿粉末对RhB和SRB的吸附越容易进行,可能是由于在同一浓度下,温度越高,RhB和SRB分子运动越剧烈,增大了与黄铁矿的碰撞概率,使吸附量增大 [13] ,由此可知,升高温度有助于黄铁矿对RhB和SRB的吸附。另外,
为正值,表明溶液中随着黄铁矿粉末吸附RhB和SRB的反应向平衡移动,固液接触面的无序性随着吸附的进行不断增大,最后达到吸附和脱附的动态平衡 [12] 。

Table 3. Thermodynamic parameters at different temperatures
表3. 不同温度下的热力学参数
3.5. 溶液pH对吸附过程的影响
溶液pH (3.0~10.0)对吸附过程的影响见图7。pH为4.0时,黄铁矿对RhB的吸附达到最大,pH为4.0~7.0时吸附量依次减小,当溶液pH继续增大(>7.0),吸附量又逐渐增大。RhB是一种阳离子染料,具有酰胺基(−NHR2)和羧基(−COOH),由于这种结构的特殊性使RhB分子表面的电荷取决于溶液pH。对于芳香羧基,pKa为4.0。当溶液pH < 4.0时,RhB分子结构中N为正价,羧基不带电荷。而黄铁矿等电点为6.4,此时黄铁矿表面带正电荷。根据静电吸引原理,阳离子染料RhB分子与黄铁矿阳离子互相排斥,导致吸附量降低。当pH > 4.0时,羧基离子化,RhB分子此时处于两性离子状态,分子表面不带电荷,而无法吸附到黄铁矿表面。当pH > 7.0时,OH−和与COO−竞争连接N+,从而导致RhB分子的聚合体减少,使吸附量增大。
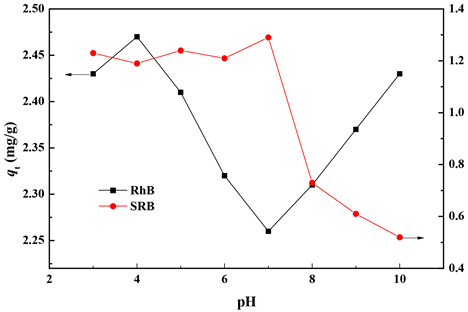
Figure 7. Adsorption of RhB and SRB by pyrite power at different pH
图7. 不同pH下黄铁矿粉末对RhB和SRB的吸附
对于SRB,结构中含有强酸性的磺酸基(−SO3H),当pH < 6.4时,SRB电离受到抑制,此时SRB分子表面不带电荷而黄铁矿表面带正电荷,因此SRB分子无法吸附到黄铁矿表面导致吸附量较小,当pH > 6.4时,SRB电离,分子表面带负电荷,而此时黄铁矿表面也带负电荷,因此吸附量较小。
4. 黄铁矿粉末再生处理
对吸附了RhB和SRB的黄铁矿粉末进行煅烧处理,以彻底去除吸附在黄铁矿表面的RhB和SRB,一方面考察黄铁矿粉末的重复利用率,另一方面对吸附后的黄铁矿粉末进行安全妥善处理避免对环境造成二次污染。收集吸附之后的黄铁矿粉末于500℃下煅烧2 h,冷却后进行重复吸附实验,结果如图8,可以看出随着煅烧次数的增多,黄铁矿粉末对RhB和SRB的吸附量都逐渐减少,煅烧六次后,黄铁矿粉末几乎无吸附作用,说明吸附了RhB和SRB的黄铁矿粉末没有多次重复利用的价值,可以直接进行回收填埋或用作工业建筑材料。
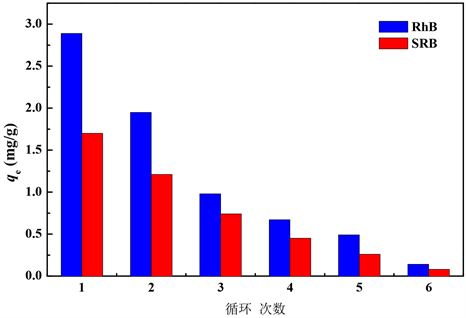
Figure 8. Cyclic adsorption of RhB and SRB on pyrite powder
图8. 黄铁矿粉末对RhB和SRB的循环吸附
5. 结论
黄铁矿对RhB和SRB的最大吸附量分别为2.89和1.70 mg/g。吸附过程都符合准二级动力学模型,理论最大饱和吸附量分别为3.04和1.78 mg/g,接近实验值;黄铁矿粉末对RhB和SRB的吸附过程都与Langumir模型拟合程度更高;升高温度有利于黄铁矿对RhB和SRB的吸附。吸附后经过两次煅烧对RhB和SRB的吸附分别为初始的67%、73%,三次煅烧后则低于40%,因此不具备多次重复利用的价值。
基金项目
湖北省自然科学基金青年项目(2016CFB190),河湖生态修复与藻类利用湖北省重点实验室开放基金(HHKF201510),湖北工业大学科研启动基金(BSQD14011),广东省水利科技创新项目(2017-14),湖北工业大学大学生创新创业训练计划:“改性粉煤灰净化富营养化水体中磷的创新实验研究” (201810500054)。
NOTES
*通讯作者。